![SOLVED: Determine the overall order of the reaction from the following data: Experiment [NO] (M) [Cl2] (M) Rate (M/s) 1 0.0300 0.0100 3.4 × 10-4 2 0.0150 0.0100 8.5 × 10-5 3 0.0150 0.0400 3.4 × 10-4 SOLVED: Determine the overall order of the reaction from the following data: Experiment [NO] (M) [Cl2] (M) Rate (M/s) 1 0.0300 0.0100 3.4 × 10-4 2 0.0150 0.0100 8.5 × 10-5 3 0.0150 0.0400 3.4 × 10-4](https://cdn.numerade.com/ask_previews/62ad3eca-8109-4f88-b404-25181f09255b_large.jpg)
SOLVED: Determine the overall order of the reaction from the following data: Experiment [NO] (M) [Cl2] (M) Rate (M/s) 1 0.0300 0.0100 3.4 × 10-4 2 0.0150 0.0100 8.5 × 10-5 3 0.0150 0.0400 3.4 × 10-4
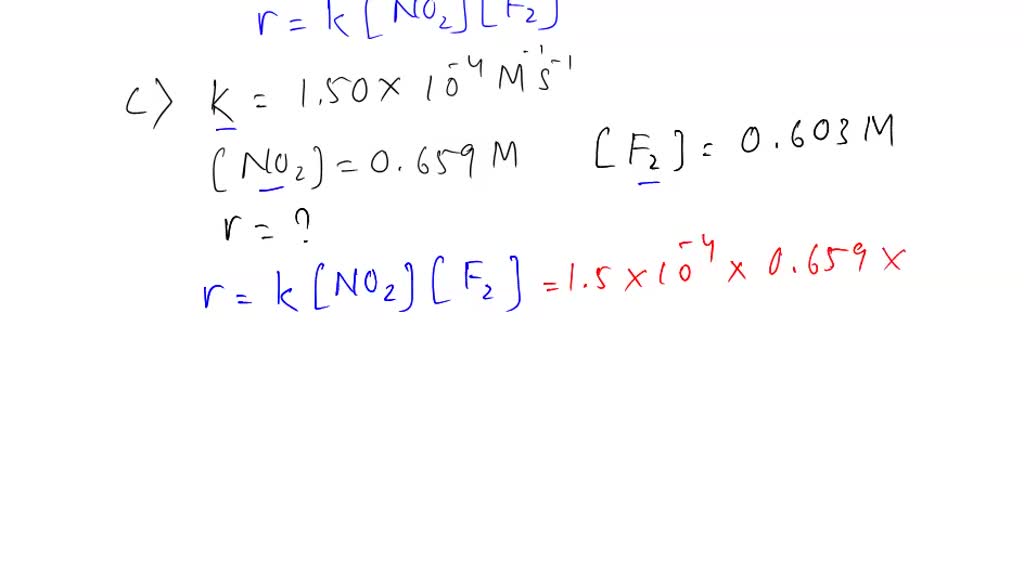
SOLVED: A. The oxidation of nitrogen monoxide by oxygen at 25 oC 2 NO + O2 2 NO2 is second order in NO and third order overall. Complete the rate law for

Amazon.com: Self Cleaning Cat Litter Box, DECEMPADS Automatic Cat Litter Box Safety Protection Extra Large Cabin Weight Sensor APP Control Timer No Scooping Smart Cat Litter Box Washable Cleaning Cabin : Pet

Amazon.com: Dickies mens Tv239 overalls and coveralls workwear apparel, Brown Duck, Large Regular US: Overalls And Coveralls Workwear Apparel: Clothing, Shoes & Jewelry
![SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]? SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]?](https://cdn.numerade.com/ask_previews/7a671ad0-bb32-4650-a5e2-0dc410a20fb1_large.jpg)
SOLVED: Classify each rate law based on whether the reaction is first-order; second-order; O third-order overall: First-order Second-order Third-order Answer Bank rate k[HCN] rate k[O] [NO] [NzE rate [BFs] [NH,] rate kINO]?
![SOLVED: Question 3 (1 point) If m=-1 and n-2,then the overall order of reaction is: Question 4 (3 points) Experiment Initial Rate (M s-1) [NOz] (M) A 0.0050 0.10 B 0.0800 0.40 SOLVED: Question 3 (1 point) If m=-1 and n-2,then the overall order of reaction is: Question 4 (3 points) Experiment Initial Rate (M s-1) [NOz] (M) A 0.0050 0.10 B 0.0800 0.40](https://cdn.numerade.com/ask_previews/80ab0b03-b365-4adf-aecd-bfc5fcfa3766_large.jpg)
SOLVED: Question 3 (1 point) If m=-1 and n-2,then the overall order of reaction is: Question 4 (3 points) Experiment Initial Rate (M s-1) [NOz] (M) A 0.0050 0.10 B 0.0800 0.40
![SOLVED:The rate law for the reaction 2 NO(g)+Cl2(g) ⟶2 NOCl(g) is given by rate =k[NO][Cl2] . (a) What is the order of the reaction? (b) A mechanism involving the following steps has SOLVED:The rate law for the reaction 2 NO(g)+Cl2(g) ⟶2 NOCl(g) is given by rate =k[NO][Cl2] . (a) What is the order of the reaction? (b) A mechanism involving the following steps has](https://cdn.numerade.com/previews/64b9f01c-0b46-4746-8320-9898e449f1a7_large.jpg)








![16.1 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube 16.1 Rate constant, overall order of reaction, order of reaction [HL IB Chemistry] - YouTube](https://i.ytimg.com/vi/9sMFJMuZzmg/maxresdefault.jpg)




