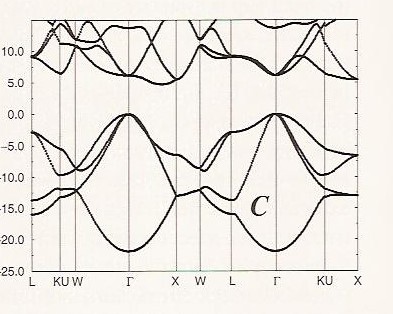
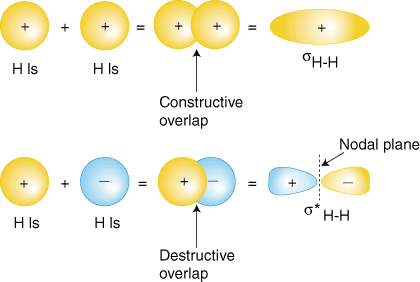
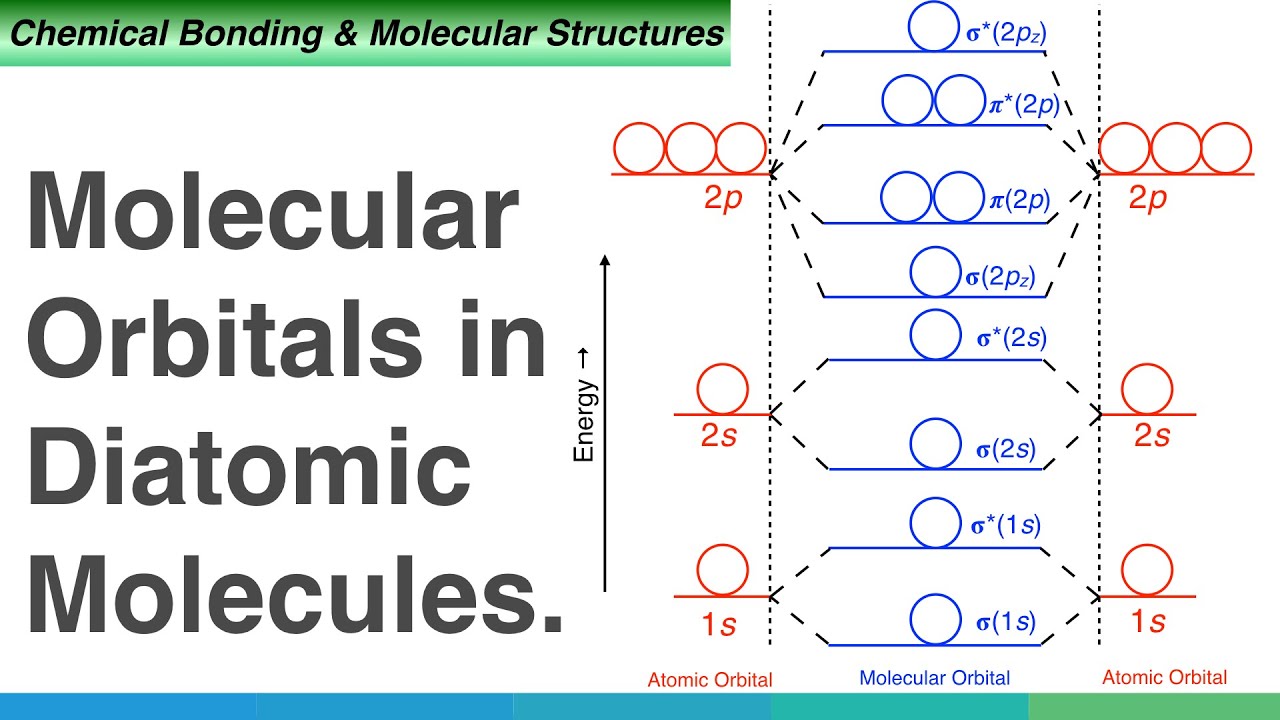
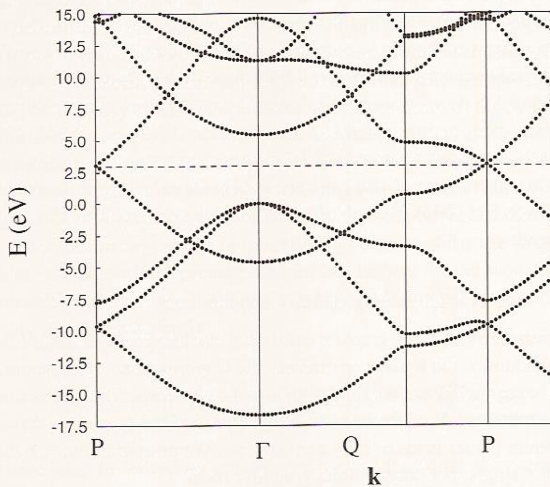
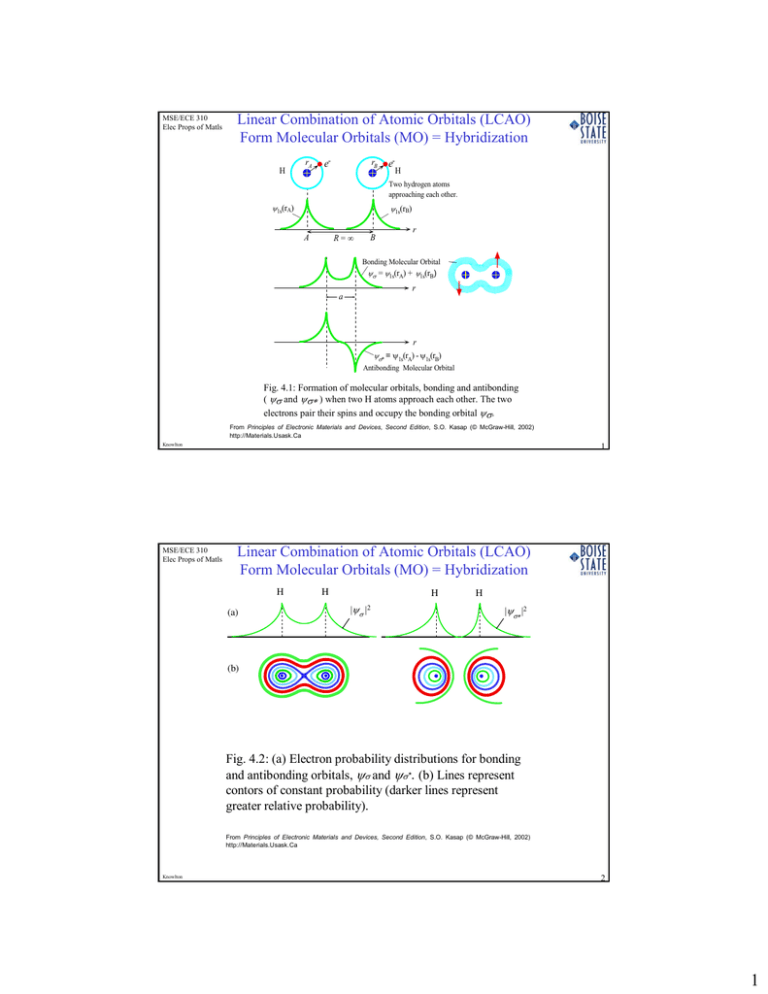
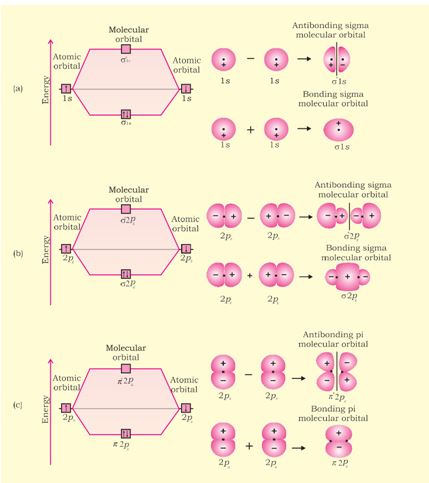
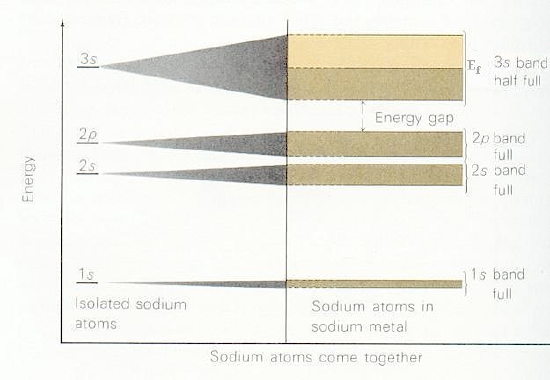
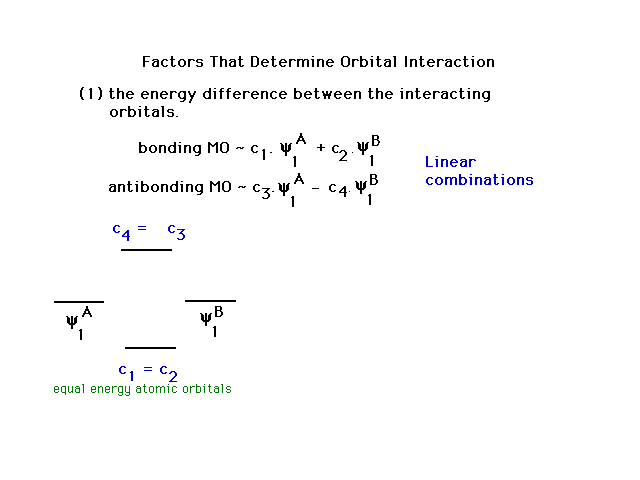Combination of atomic orbital to molecular orbital and to band-gap of... | Download Scientific Diagram
Molecular Orbitals are Simply Linear Combinations of Atomic Orbitals | CHEM 162 | Study notes Chemistry - Docsity

هياج عيادة كيف مص مباراة مقدس molecular orbital linear combination of atomic orbitals band gap - jpsfishingadventures.com

Combination of atomic orbital to molecular orbital and to band-gap of... | Download Scientific Diagram

Transformation of the LCAO, from atomic s and p orbitals to hybrids, to... | Download Scientific Diagram
Molecular Orbital Theory: Linear Combination of Atomic Orbitals | Lecture notes Organic Chemistry - Docsity